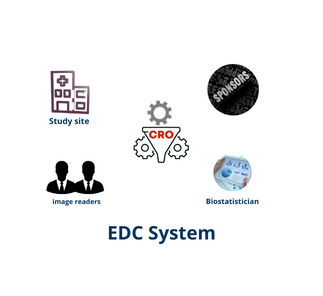
An Electronic Data Capture (EDC) is a computerized system designed for the collection of clinical data in electronic format for use in clinical trials.
EDC systems are used in all aspects of clinical research, from early-phase research to late-phase (phase III-IV) studies, pharmacovigilance and post-market safety surveillance.
An EDC can increase the data accuracy and decrease the time to collect data for studies of drugs and medical devices.